Clinical Resources
Materials for Providers
Alphabetical by Title
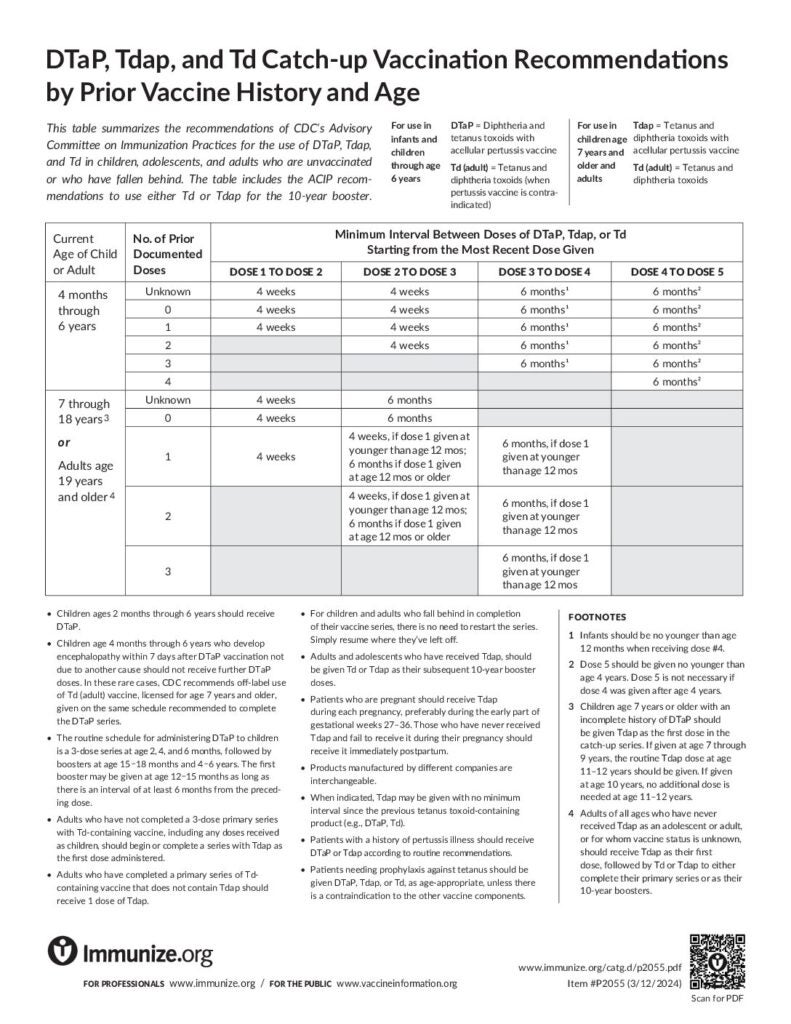
DTaP, Tdap, and Td Catch-Up Vaccination Recommendations by Prior Vaccine History and Age
This table summarizes the catch-up recommendations of CDC’s ACIP for the use of DTaP, Tdap, and Td vaccines.
Standing Orders for Administering Diphtheria, Tetanus, and Acellular Pertussis (DTaP) Vaccine to Children Younger Than Age 7 Years
Eligible healthcare professionals may vaccinate children under 7 who meet any of the criteria on this form. This template is consistent with the American Academy of Pediatrics 2026 Recommended Child and Adolescent Immunization Schedule.
Standing Orders for Administering Td/Tdap Vaccine to Adults
Eligible healthcare professionals may vaccinate adults who meet any of the criteria on this form
Standing Orders for Administering Tdap During Pregnancy
Eligible healthcare professionals may vaccinate all pregnant women who meet any of the criteria on this form. This template is consistent with recommendations of the American Academy of Pediatrics and the American College of Obstetricians and Gynecologists.
Standing Orders for Administering Tdap/Td Vaccine to Children and Teens Age 7 Years and Older
Eligible healthcare professionals may vaccinate children and teens who meet any of the criteria on this form. This template is consistent with the American Academy of Pediatrics 2026 Recommended Child and Adolescent Immunization Schedule.
Ask the Experts
CDC · FDA · State
ACIP Recommendations
Current Recommendations
Additional Federal Resources
- All current and archived ACIP Tetanus recommendations
- General Best Practice Guidelines for Immunization
- ACIP Tetanus recommendations at CDC
- Diphtheria, Tetanus, and Pertussis Vaccination: Information for Healthcare Providers
- About Diphtheria, Tetanus, and Pertussis Vaccines
- Tetanus Vaccine Recommendations
CDC Recommended Schedules
FDA Package Inserts & EUAs
State Policies
Travel
All travelers should be up to date on routine vaccines. Depending on the destination, itinerary, and duration of travel, additional vaccines may be recommended.
CDC Resources
Travelers’ health information for healthcare providers