Clinical Resources
Materials for Providers
Alphabetical by Title
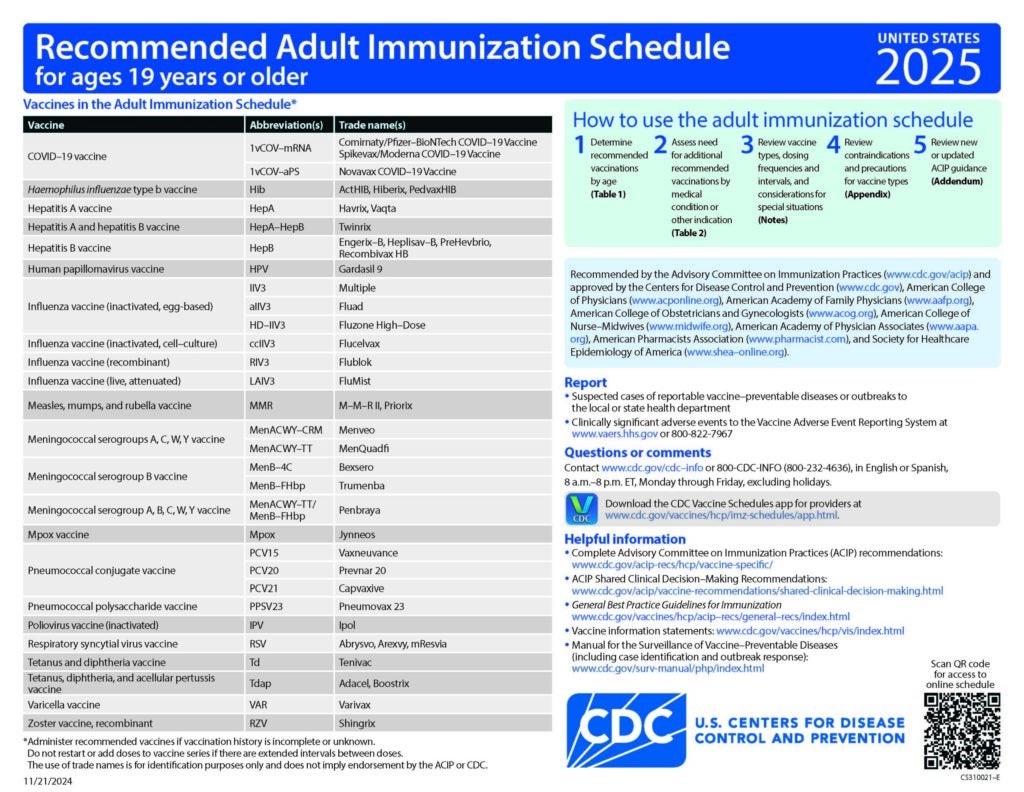
Meningococcal B Vaccine Recommendations by Age and Risk Factor
This piece gives recommendations for MenB vaccine use in people age 10 years or older.
Standing Orders for Administering Meningococcal B Vaccine (MenB) to Adolescents and Adults
Eligible healthcare professionals may vaccinate adolescents and adults who meet any of the criteria on this form. This template is consistent with the American Academy of Pediatrics 2026 Recommended Child and Adolescent Immunization Schedule.
CDC · FDA · State
ACIP Recommendations
Current Recommendations
Additional Federal Resources
CDC Recommended Schedules
FDA Package Inserts & EUAs
Healthcare Professional Organizations
More from Immunize.org
Unprotected People Stories
Photos & Videos
Immunize.org’s MenB Vaccination Honor Roll
Preventing Outbreaks of Meningitis B in Colleges and Universities
Immunize.org urges colleges and universities to establish policies requiring or recommending meningococcal serogroup B (MenB) vaccination to protect their students and prevent outbreaks. Immunize.org’s MenB Vaccination Honor Roll recognizes champion institutions that have taken the lead in establishing such policies.